*All other brand or product names located in this site are Trademarks of their respective holders.
Bortezomib for Injection
NDC number :
60505-6050-04
Material number :
63996
Active ingredient :
Bortezomib
Bioequivalent to * :
VELCADE®
Therapeutic class (AHFS) :
Antineoplastic Agents
Strength :
3.5MG
Pack size (form) :
10ML INJECTABLE (VIAL)
Dosage form :
Injectable Single Dose
Scored :
N/A
Color :
White to off-white
Shape :
N/A
Markings :
N/A
Route of administration :
Subcutaneous, Intravenous
Storage condition :
Controlled Room Temperature (68 - 77 degrees F)
Minimum order quantity :
50
Multiple order quantity :
50
Latex free :
Yes
Dye free :
Yes
Preservative free :
Yes
Hazardous material :
Yes
Product resources :
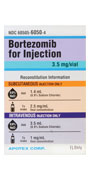 not to scale
not to scalePackaging information :
| Length | Width | Height | Weight | Units/Pack | |
|---|---|---|---|---|---|
| Unit | 2.244 in | 1.575 in | 3.543 in | 0.088 lb | N/A |
| Inner | 0 in | 0 in | 0 in | 0 lb | 1 |
| Case | 11.811 in | 8.268 in | 7.677 in | 6.393 lb | 50 |
